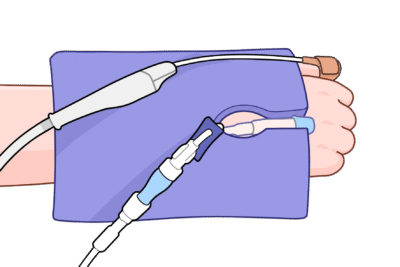
Introducing the EcoPad!
Prioritizing excellent patient protection, easy set-up, and environmental sustainability.
Proudly Made in the USA from foam manufacturing to fabrication.
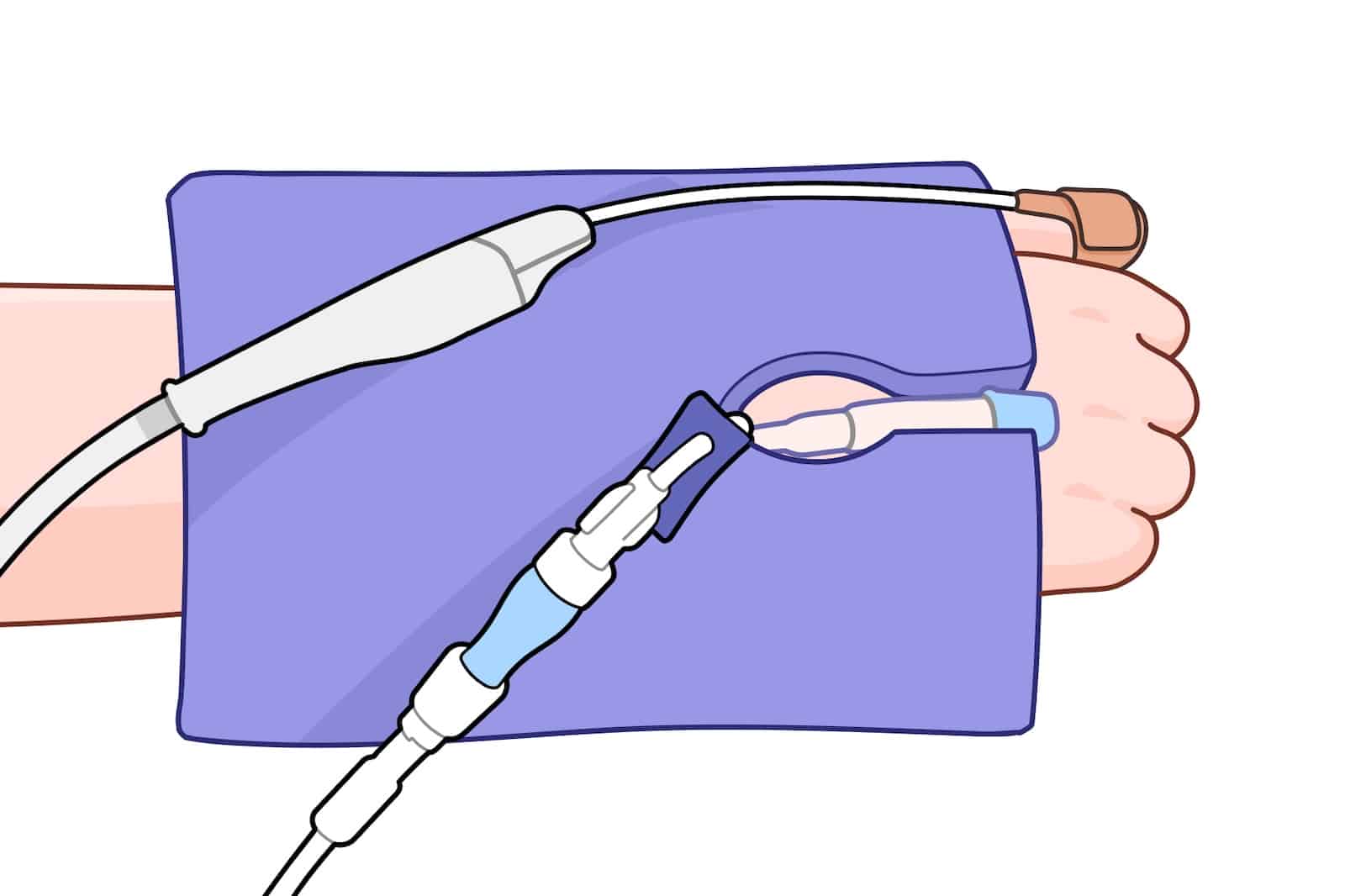
Cushion Device Pressure—And Route Lines Cleanly When Needed
For patients under anesthesia, sedation, or limited mobility, hard device and equipment surfaces (connectors, housings, brackets, edges) can rest against skin for hours—especially at bony prominences—increasing pressure, friction, and shear.
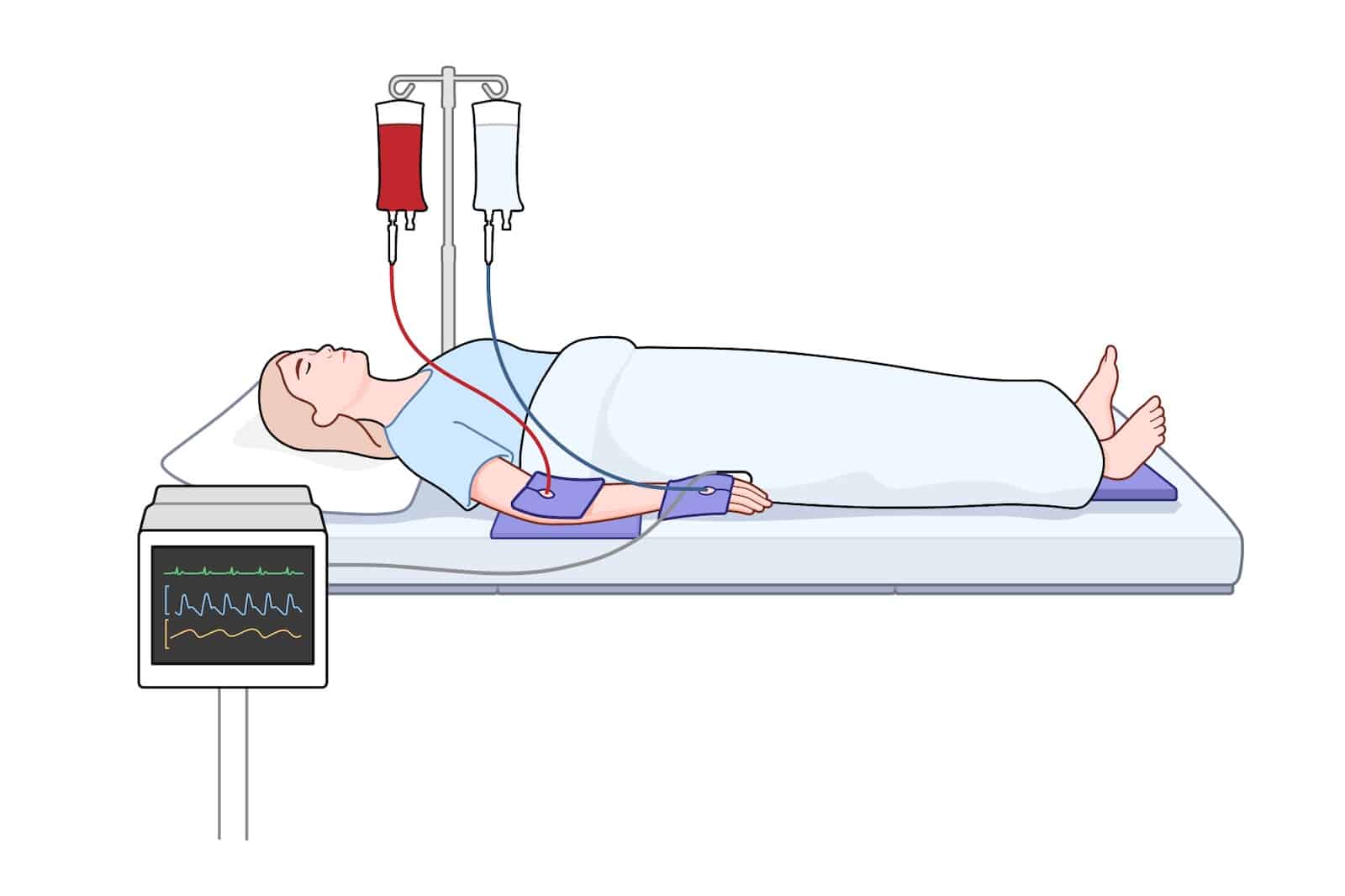
The EcoPad provides versatile cushioning between skin and devices across care settings.
Available in two easy to use sizes:
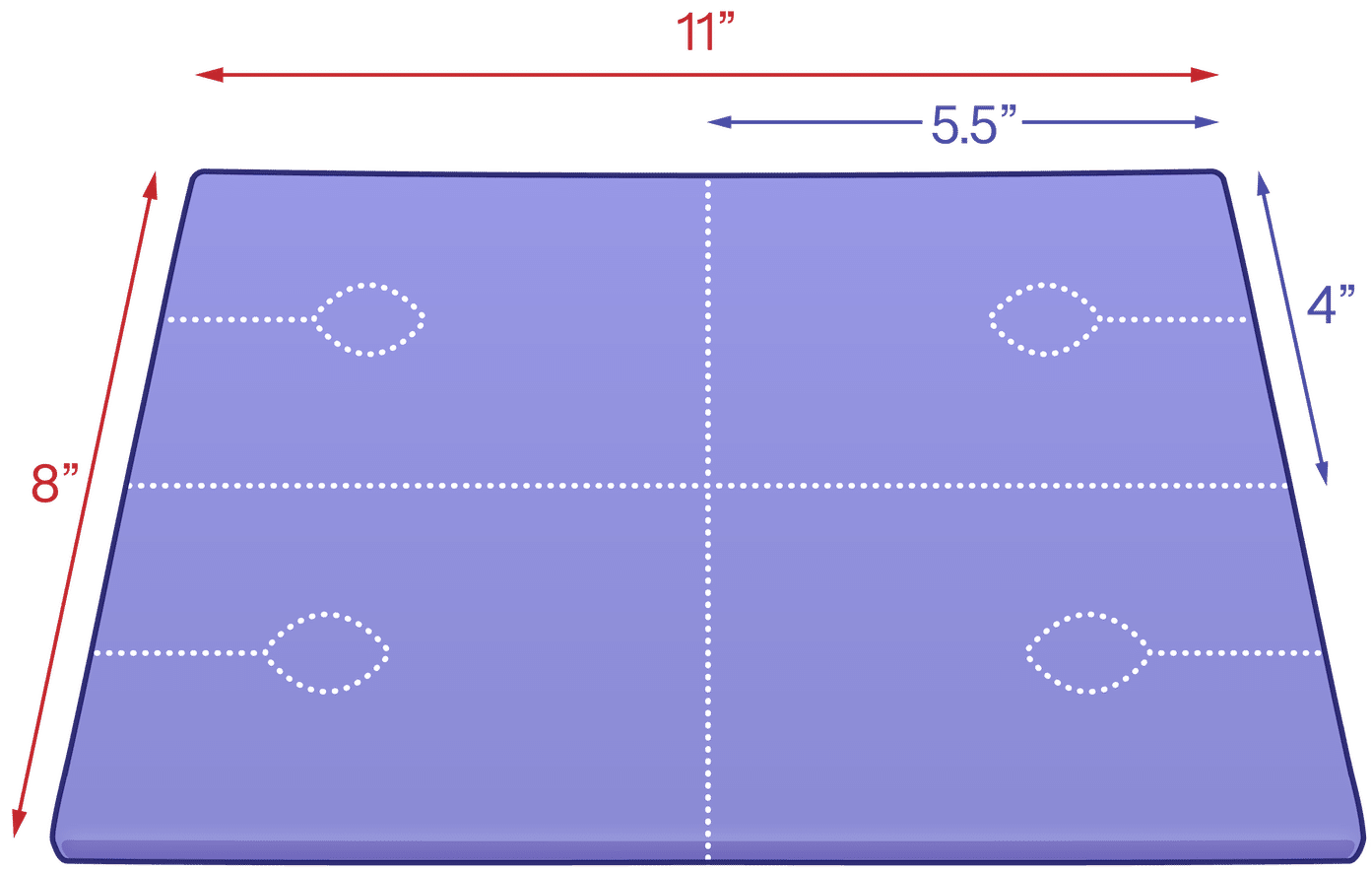
EcoPad Small IV: 0.5″ x 8″ x 11″‘
When lines or tubes are present, the perforated slit + 1″ central port let you route them cleanly without kinking, while the surrounding foam helps off-load contact pressure from adjacent components.
EcoPad Large: 1″ x 10″ x 15″
For the applications that require a little extra padding to reduce pressure related injuries and improve patient comfort. Use to cushion bony or other high pressure areas with a pad that has a thick, broad cushion area.
Clinical Insight & Standards
Build in Protection—Don’t Rely on “It’ll Be Fine”
AORN Guidelines for Perioperative Practice emphasize verifying that devices and equipment do not rest directly against the patient, and recommend applying prophylactic materials to bony prominences and other pressure‑prone areas. (AORN Guidelines for Perioperative Practice.)
The EcoPad makes this protection simple and repeatable: the slit‑and‑port design routes the line without deformation while the surrounding foam off‑loads edges and connectors, helping reduce focal pressure, friction, and shear.
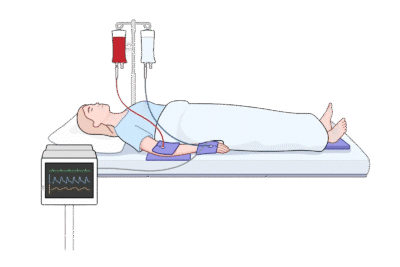
Ditch the Makeshift Fixes—Support Consistency and Compliance
Ad‑hoc cutting or tearing of generic foam blocks is time‑consuming, variable, and can shed particles. Improvised positioning aids are discouraged by modern perioperative guidance and can expose teams to quality, safety, and survey findings. (AORN Guidelines for Perioperative Practice – Positioning the Patient – Section 2.9.1)
The EcoPad replaces improvised workarounds with a purpose‑built, clean‑tear solution that deploys in seconds and looks professional at the bedside.
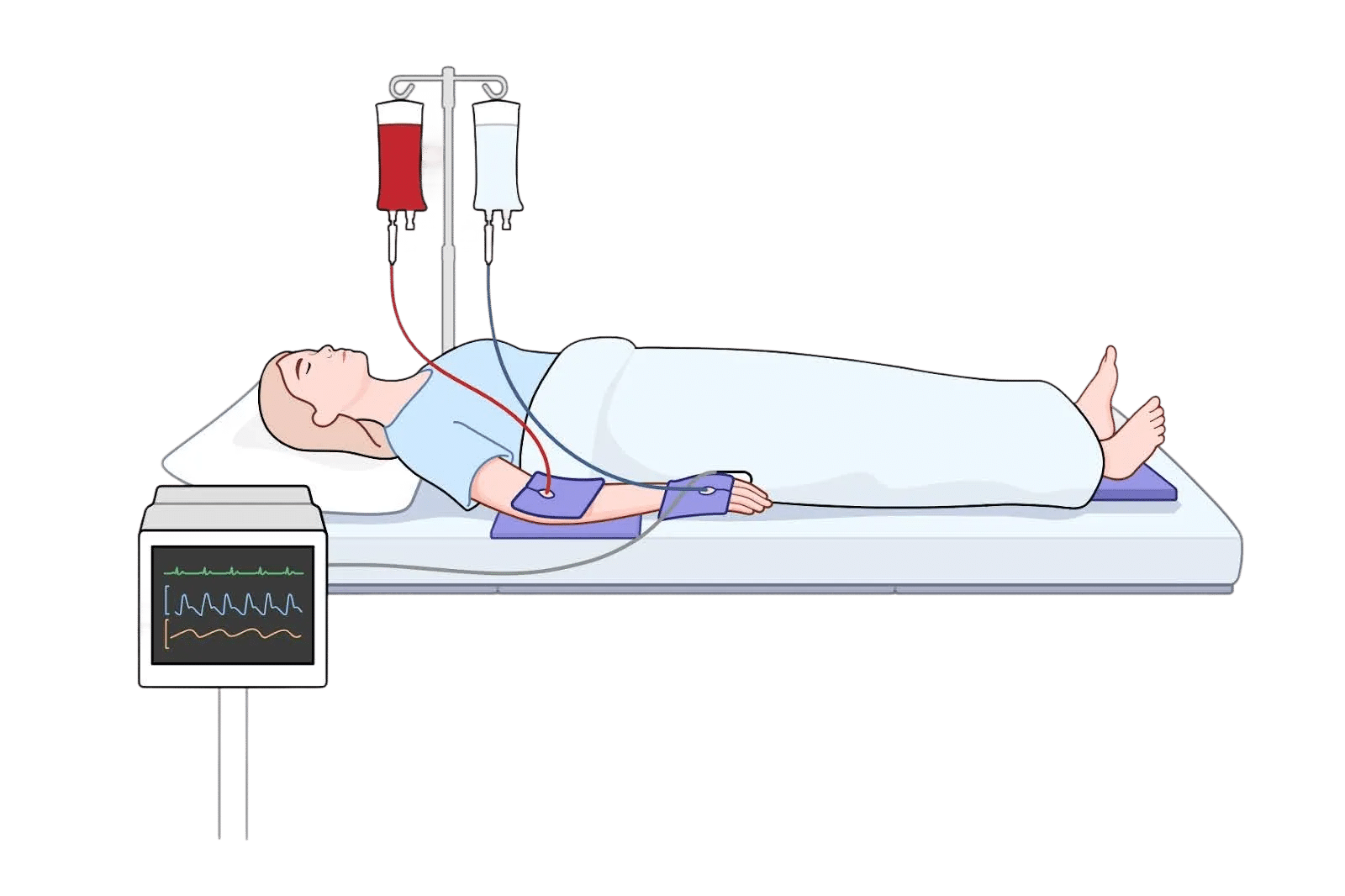
Essential Near High‑Risk Anatomy
Bony prominences and thinly padded regions (such as the wrist, elbow, ankle…) are more vulnerable to pressure and shear. The pad’s 0.5 in (Small IV) or 1 in (Large) thickness offers a consistent buffer that can be tailored to anatomy and device layout.

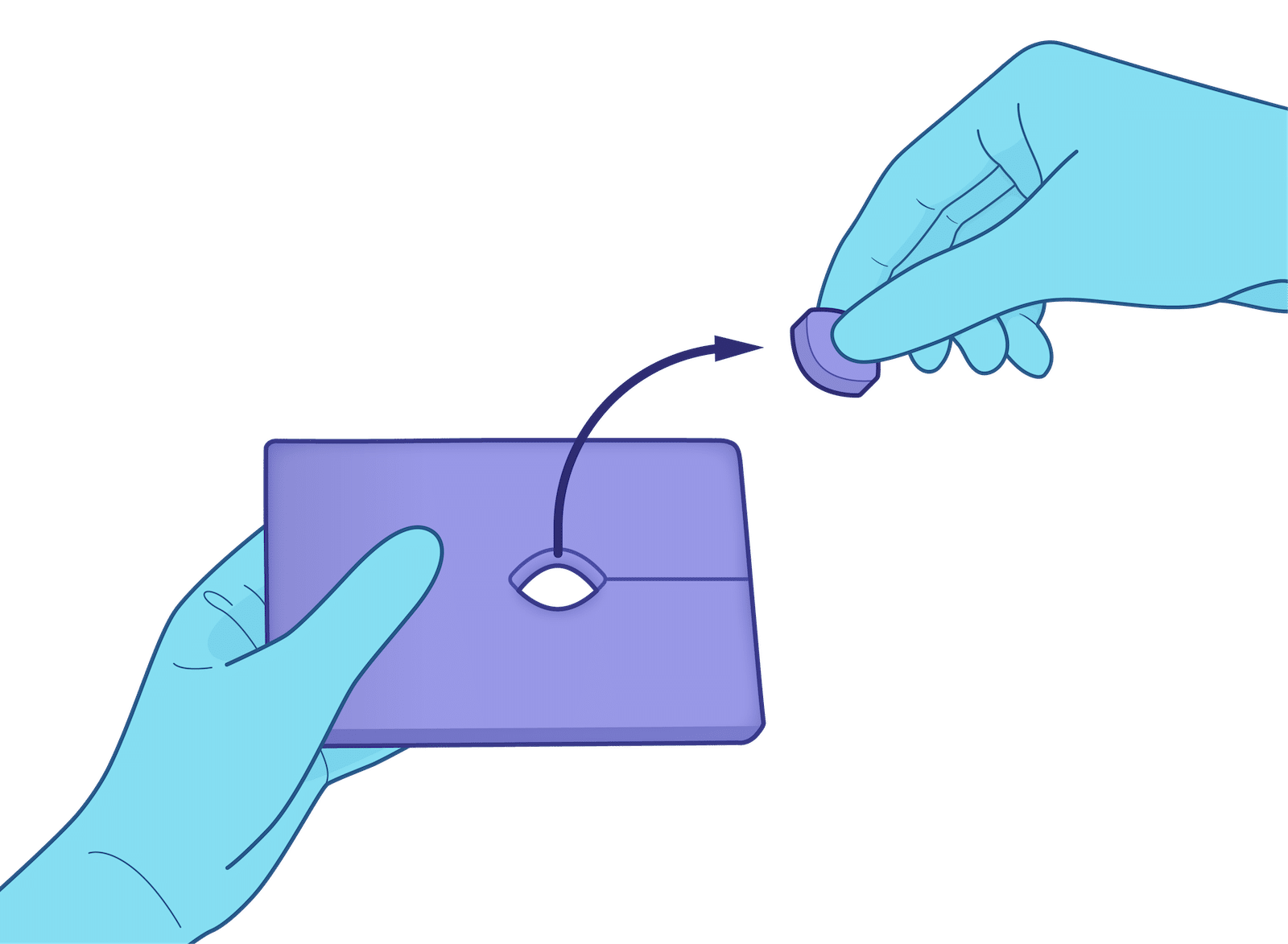

Eco-Pad Small IV: Faster Setup. Better Protection. More Focus on the Patient.
-
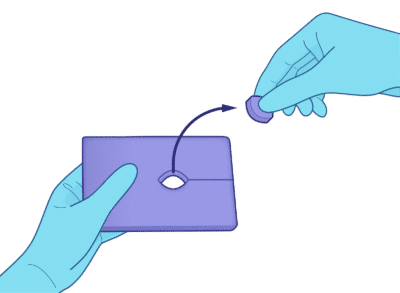
No tools required — Tear along the slit perforation and pop out the center port in seconds.
-
Place, route, and position — Slip the device line through the slit into the port; rest connectors on the pad, not skin.
-
Stable, repeatable setups — Reduce re‑positioning and readjustment compared to makeshift methods.
Sustainability without Compromising Care
Healthcare generates significant solid waste—foams included. The EcoPad’s biodegradable polyurethane (tested to ASTM D5511) supports initiatives to reduce landfill burden while preserving the convenience of single‑patient use.
The EcoPad Small IV Advantage:
Designed by Clinicians. Built for Versatile Care.
1. Skin‑Friendly Foam
Breathable, soft, and comfortable; non‑adhesive to help minimize skin trauma during removal.
2. Clean Tear, Professional Look
Purpose‑made perforations reduce particle shedding vs. ad‑hoc cuts and help avoid makeshift appearance at bedside.

3. Perforated Access Slit (1.5 in)
Opens on demand from pad edge to center. No scissors or scalpels needed.
4. 1 in Central Port
Provides a clean, repeatable pathway for lines/tubes without crimping; maintains device geometry.
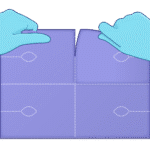
Step 1
Separate one 4 × 5.5 in pad along the sheet perforations.
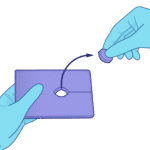
Step 2
Create the port by gently tearing along the 1.5 in perforation and popping out the 1 in center circle.
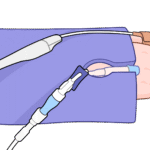
Step 3
Route the device: Guide the line through the slit into the central port.
Position and check: Place connectors and hard edges on the pad, away from skin; confirm there is no device resting directly on the patient.
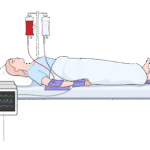
Step 4
Monitor per protocol and replace pad as needed.
Why Teams Choose the EcoPad
- Versatile cushioning for device‑to‑skin interfaces.
- Purpose‑built slit + port for clean line routing—no scissors or scalpels.
- Fast, clean setup that looks professional at bedside and in the OR.
- Supports compliance with perioperative positioning and pressure‑injury prevention guidance.
- Comfortable for patients: breathable, soft, and non‑adhesive.
- Sustainability‑minded: biodegradable polyurethane (ASTM D5511).
- Made in the USA.
How to Order
To place an order, set up an invoicing account, or ask about distribution opportunities, contact us directly. Our team will assist you with purchasing and answer any questions.
or call us at 330-247-5805 to place an order.